Welcome to the blog of the project MetClimVOC!
June 2022
During the lifecycle of a research project, valuable data are generated. How should this research data be managed? What happens with the data once the project is finished? You will find more about research data management and data sharing on this month post written by our guests.
# Let’s talk about research data management
The project MetClimVOC is collaborating with the project TC-IM 1449 in order to establish good practices for the organisation, documentation, conservation and dissemination of all data handled within a research project in general. All these activities are known under the umbrella term “Research Data Management”(RDM).
The core idea behind current RDM is that the results of scientific activities shall be made findable, accessible, interoperable and re-usable (Figure 1)– independently if they are made available to the public (open data), or just conserved (archived) for internal use. This very concise criteria catalogue comprises more detailed requirements concerning file formats, metadata, licenses, identifiers, etc.
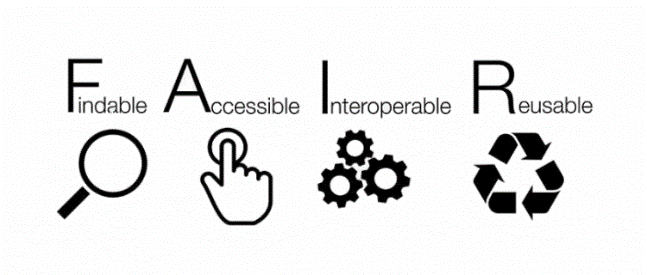
# What does it mean to be FAIR?
The FAIR data principles are defined by the GO FAIR Initiative:
Findability refers to the capability of data to be located both by humans and by computers. Main requirements on this first step are rich, indexed and machine-readable metadata as an essential component for automatic discovery of datasets and services.
Accessibility refers to the possibility to get the data once found, possibly upon authentication and authorisation. Besides machine readability, this step requires an open communication protocol as well as persistency of address and metadata.
Interoperability of data refers to their capability to be integrated with other data, as well as with applications and/or workflows for further functions, such as analysis, storage, and processing. A condition for that is the usage of standard vocabularies and representation languages and correct qualified referencing to other (meta)data.
Reusability of data is the ultimate goal of data FAIRness. To achieve this, both metadata and data should be richly described conform to domain-relevant community standards, contain detailed provenance information, and be offered with a clear and accessible usage license, so that they can be replicated and/or combined in different settings.
These principles can be applied and evaluated at different levels: 1) single datasets (or any digital object), 2) metadata (i.e., information about that digital object), and 3) infrastructure. More in-depth information about practical implementation of the FAIR data principles is provided by the GO FAIR Initiative.
# Where to start?
The most important activities connected with RDM (Figure 2) are:
- - Data organisation and protection (e.g., choice of suitable work platforms, naming conventions)
- - Long-term storage (publication on a data repository or internal storage in an archive).
- - Planning of resources and tasks (Data Management Plan (DMP).
- - Documentation of the research work (via a lab notebook or via annotation of the data files).
- - Legal aspects may also play a role in RDM.
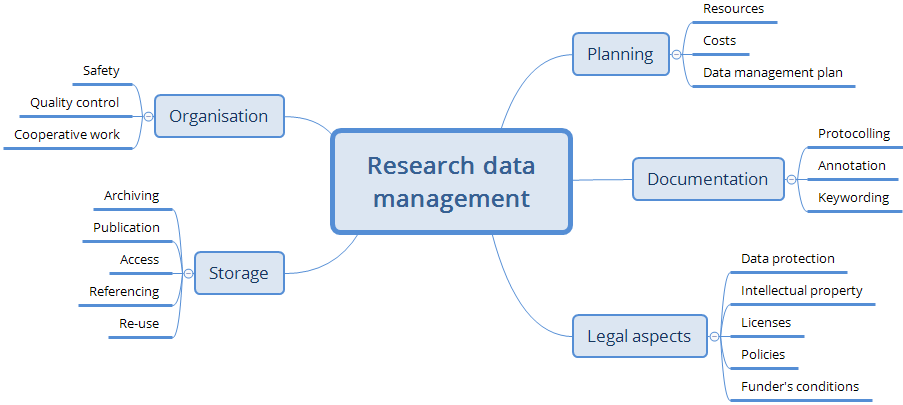
Most of these tasks can be performed with the help of dedicated software tools, which allow propagating the data and related information (metadata) along the research workflow.
A structured RDM goes further beyond the obligations set by the funding organisations (data management plan, data publication): with the help of proper tools, guidance and good practice examples researchers can manage their data in a more effective and sustainable way, with benefit for themselves and for the whole scientific community.
# TC-IM 1449: specialised support for RDM in metrology
The project TC-IM 1449 “Research Data Management and the European Open Science Cloud” has been initiated to improve and strengthen the involvement of the metrological community in topics concerning RDM by increasing RDM awareness and skills among researchers, on the one hand. On the other hand, by bringing metrological data quality principles - such as traceability, uncertainty propagation, reproducibility and trustworthiness - into the current RDM / FAIR discourse and into leading initiatives such as the European Open Science Cloud (EOSC).
The TC-IM 1449 project involves scientists, librarians and IT and digitalisation experts from 12 European National Metrology Institutes and is part of the EURAMET Technical Committee for Interdisciplinary Metrology (TC-IM). The following actions are ongoing since its start in June 2018:
- - Elaboration of RDM guidelines and checklists for researchers.
- - Elaboration of sample reporting documents and RDM policies.
- - Definition of minimum metadata requirements and standards for metrological information.
- - Motivation of the researchers and dissemination of best practices.
In this context, the interaction with MetClimVOC has offered TC-IM 1449 the possibility to follow a project from its beginning (i.e., early stages of the implementation) to its end (i.e., preservation and reporting). This will be an essential contribution in the deployment of good practice examples and will deliver valuable input for material such as checklists, guidelines and templates for future researchers dealing with Data Management Plans and Research Data Management in general.
The TC-IM 1449 project seeks to increase its representativeness within the European metrological landscape. Therefore, anybody who wants to engage is warmly welcome to join the project by contacting the project coordinator, Giacomo Lanza (PTB, Germany).
#About the authors
Giacomo Lanza, coordinator for research data management at PTB's Library. His main action fields are the interaction of metrology and RDM, development of standards, metadata and terminologies, and generation and implementation of DMP for European projects. He is active in the German initiative for a National Research Data Infrastructure (NFDI) and in the user community of the Research Data Management Organiser (RDMO). In his previous employment as a researcher, he dealt with the balance of greenhouse gases in agriculture and animal husbandry and previously with optical spectroscopy of intrinsic fluorescent proteins.

Federico Grasso Toro, project leader in the Research and Development (R+D) at METAS. His activities in EURAMET include TC-IM 1448 project for "Development of digital calibration certificates", TC-IM 1449 project for "Research data management and the European Open Science Cloud" and the newly formed project TC-IM 1551 for "Challenges and opportunities in sensor network metrology", from which he is the coordinator.
#Previous posts
In this section, you can find the links to previous blog posts.
February 2022 - Reference gas mixtures for oxy-VOCs
December 2021 - Terpenes in the atmosphere
October 2021 - Monitoring halogenated VOCs from the space
June 2021 - One year of project MetClimVOC!
March 2021 - How gas surface interactions can affect VOC monitoring?
January 2021 - Halogenated VOCs in the atmosphere: monitoring and traceability
November 2020 - Atmospheric trace gases: VOCs, impact climate change
September 2020 - Welcome to the blog of the project MetClimVOC!